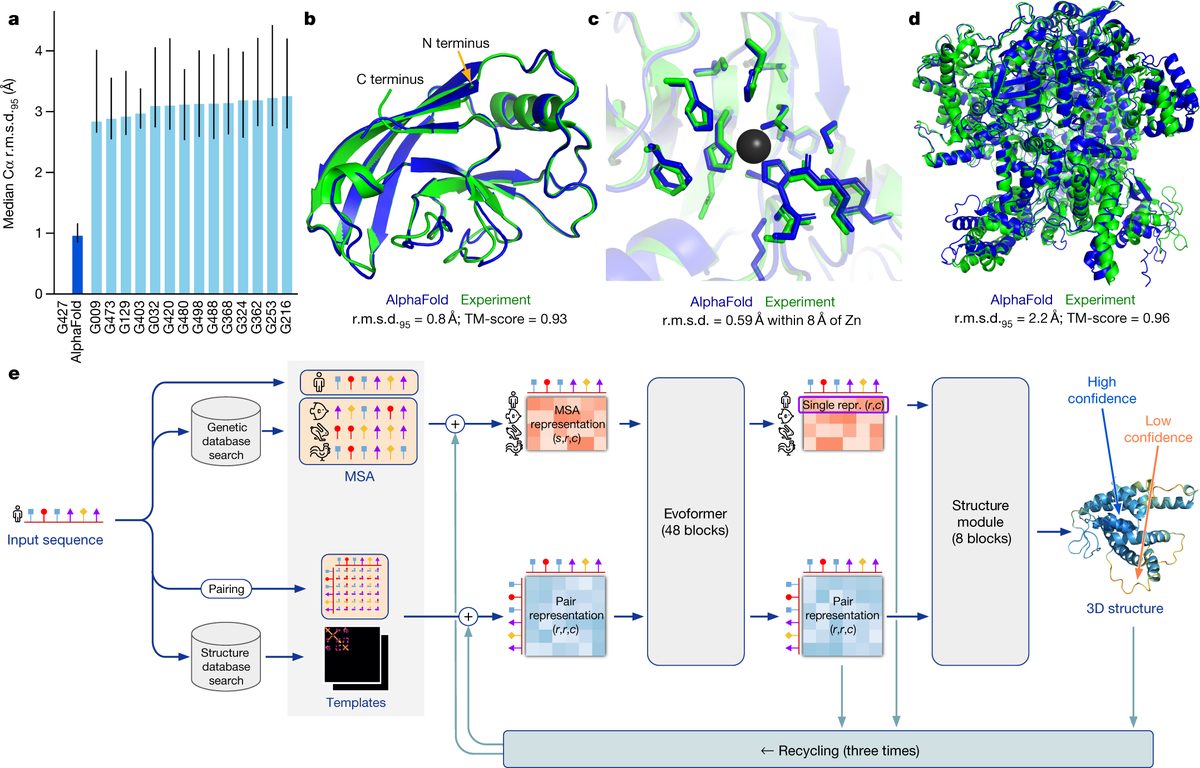
Human Genome Project (1990-2003)
1990-2003What Happened
A 13-year, $2.7 billion international effort sequenced the roughly 3 billion base pairs of human DNA. The project competed with Craig Venter's privately funded Celera Genomics, which used a faster shotgun sequencing method and announced a draft genome simultaneously with the public effort in 2000.
Outcome
Both the public consortium and Celera published draft genomes in February 2001. The public data was made freely available, while Celera initially restricted access through a subscription model.
The open-access model won. Free genome data enabled thousands of studies that Celera's proprietary model could not have supported. The project catalyzed the genomics industry, drove DNA sequencing costs from $100 million per genome in 2001 to under $200 by 2024, and established a precedent for open science in biology.
Why It's Relevant Today
The tension between Isomorphic Labs' proprietary IsoDDE and the open AlphaFold database mirrors the Celera-versus-public-consortium dynamic. The genome project showed that open access to foundational biological data produces more aggregate innovation, a lesson that shapes the current debate over whether AI drug design tools should be public goods.