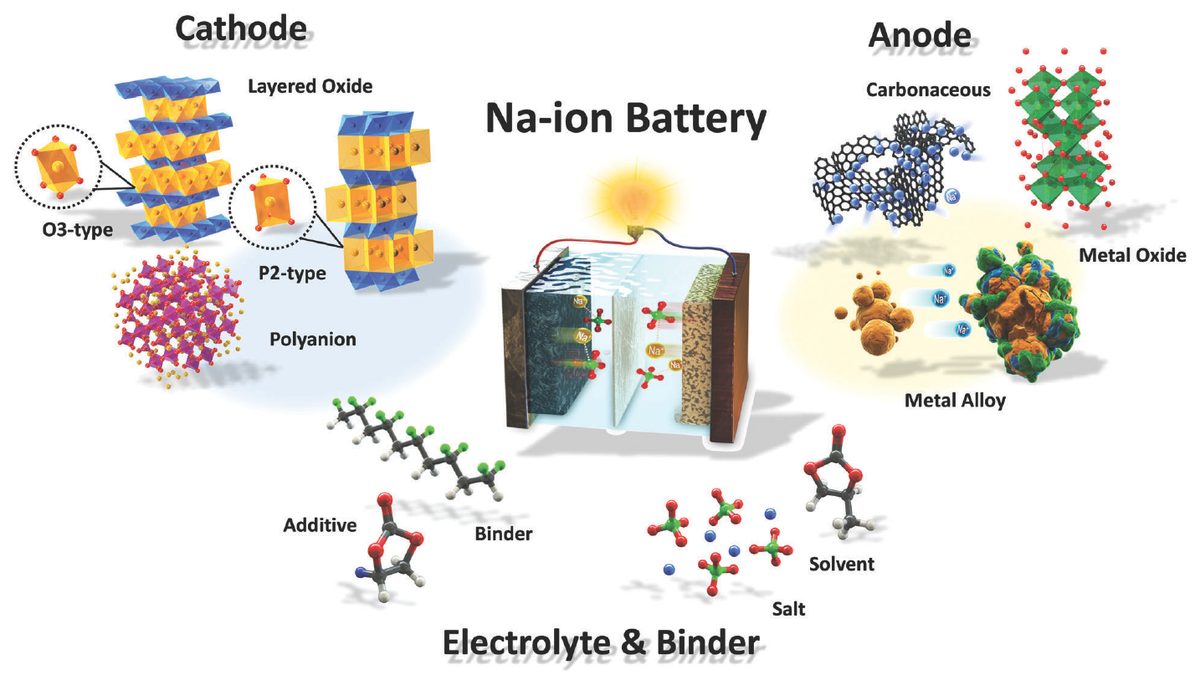
BYD Blade Battery nail-penetration test (2020)
BYD publicly demonstrated its Blade Battery, a lithium iron phosphate cell in a thin, elongated form factor, passing a nail-penetration test — the most demanding battery safety standard — without producing smoke or fire. The cell surface reached only 30-60 degrees Celsius. Conventional nickel-manganese-cobalt cells subjected to the same test burst into flames.
The demonstration made global headlines and gave BYD a powerful marketing differentiator. The Blade Battery became standard across BYD's vehicle lineup.
It proved that cell-chemistry-level safety — rather than pack-level engineering workarounds — was commercially viable and consumer-relevant, setting the benchmark that the CAS sodium-ion research now builds upon.
The CAS fireproof sodium-ion battery passes the same nail-penetration test, but using an even cheaper, more abundant chemistry. If BYD's Blade Battery showed that inherent safety sells cars, the CAS breakthrough shows it can be done without lithium at all.