Pemazyre approval for bile duct cancer (2020)
April 2020What Happened
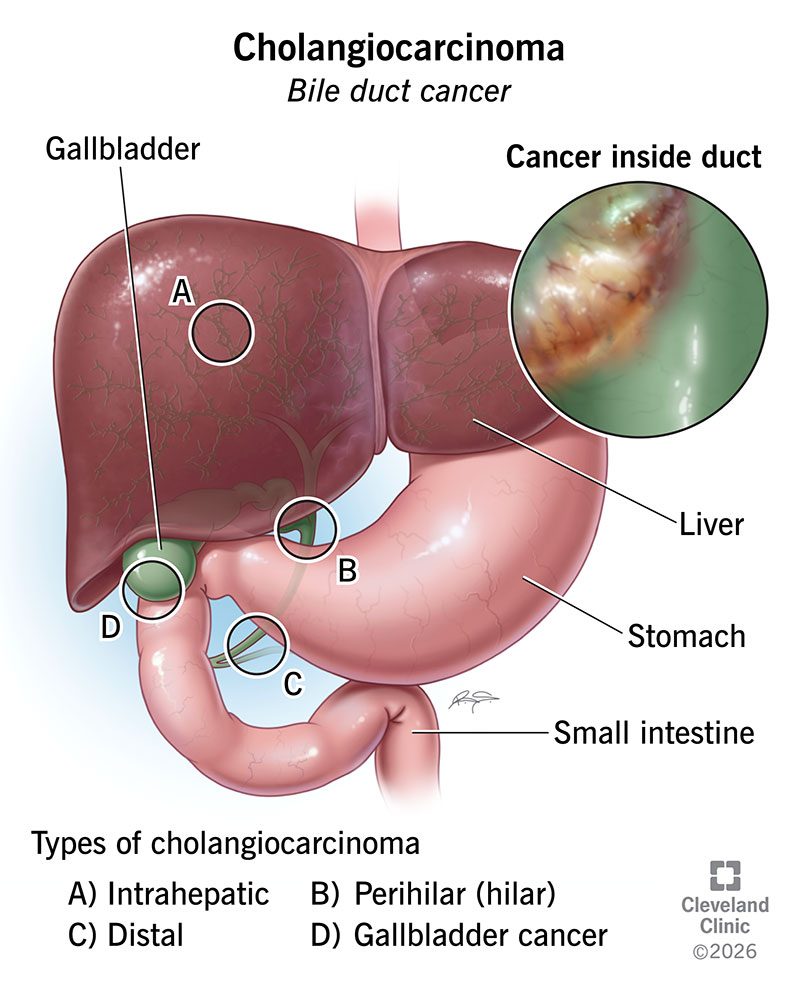
The FDA granted accelerated approval to Pemazyre (pemigatinib) for adults with previously treated cholangiocarcinoma carrying an FGFR2 fusion. It was the first targeted therapy approved for any subtype of bile duct cancer, a disease where standard chemotherapy had stalled in effectiveness for years.
Outcome
Pemazyre gave roughly 10 to 15 percent of bile duct cancer patients their first option beyond chemotherapy. Tumor shrinkage rates in trials ran near 36 percent.
The approval established cholangiocarcinoma as a disease defined by genetic subtypes rather than a single entity. It opened the door to FDA approvals for other targeted therapies in the disease, including IDH1 inhibitors and now the NRG1-targeted Bizengri.
Why It's Relevant Today
Pemazyre showed that genetic testing in bile duct cancer can find patients who respond to targeted drugs even when chemotherapy fails. Bizengri extends that model to a different, even rarer genetic subtype.