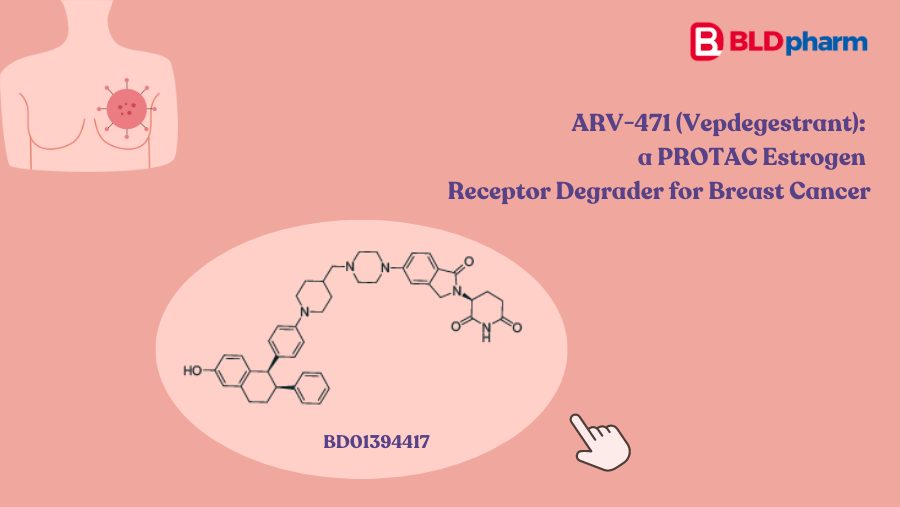
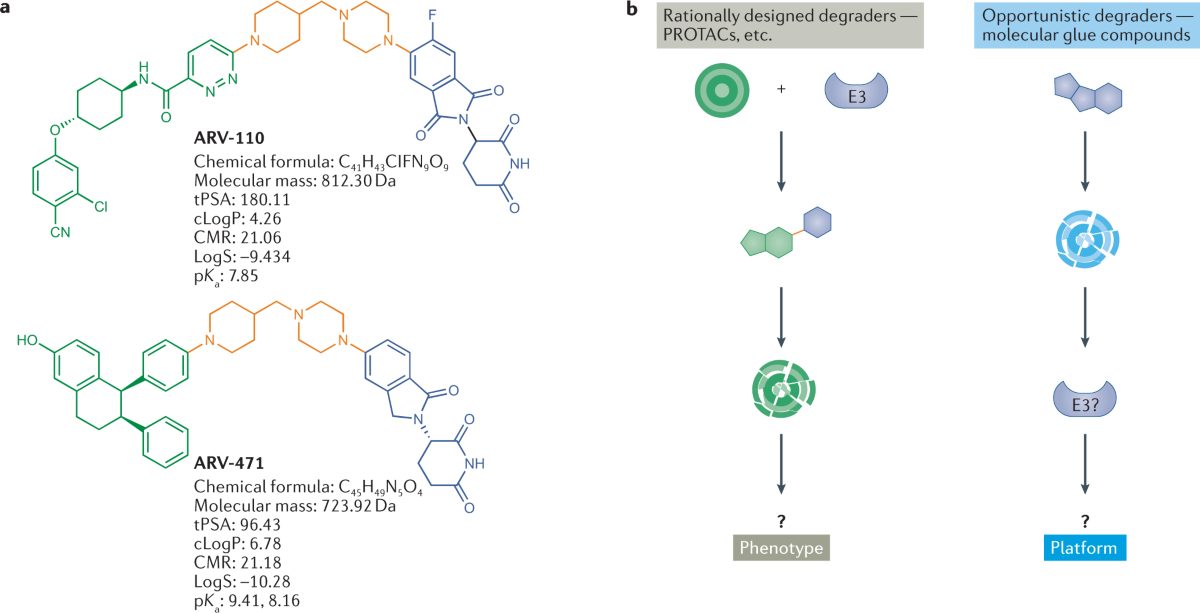
Gleevec (imatinib) approval (2001)
May 2001What Happened
FDA approved Novartis's imatinib for chronic myeloid leukemia in just 2.5 months, validating Brian Druker's argument that designing a small molecule to block a single cancer-driving kinase (BCR-ABL) could turn a fatal disease into a manageable one. The drug produced durable remissions in patients who had previously had a five-year survival of about 30%.
Outcome
Imatinib became the foundation of CML treatment and Novartis's biggest oncology product, validating targeted therapy as a paradigm.
More than 70 kinase inhibitors have since been approved across cancers, building a multi-billion-dollar drug class that did not exist before Gleevec.
Why It's Relevant Today
Like PROTACs, kinase inhibitors were a long-pursued idea waiting for a first approval to unlock investor and regulator confidence. Gleevec's success made the next dozen kinase inhibitor approvals routine — the template VEPPANU may now provide for protein degraders.