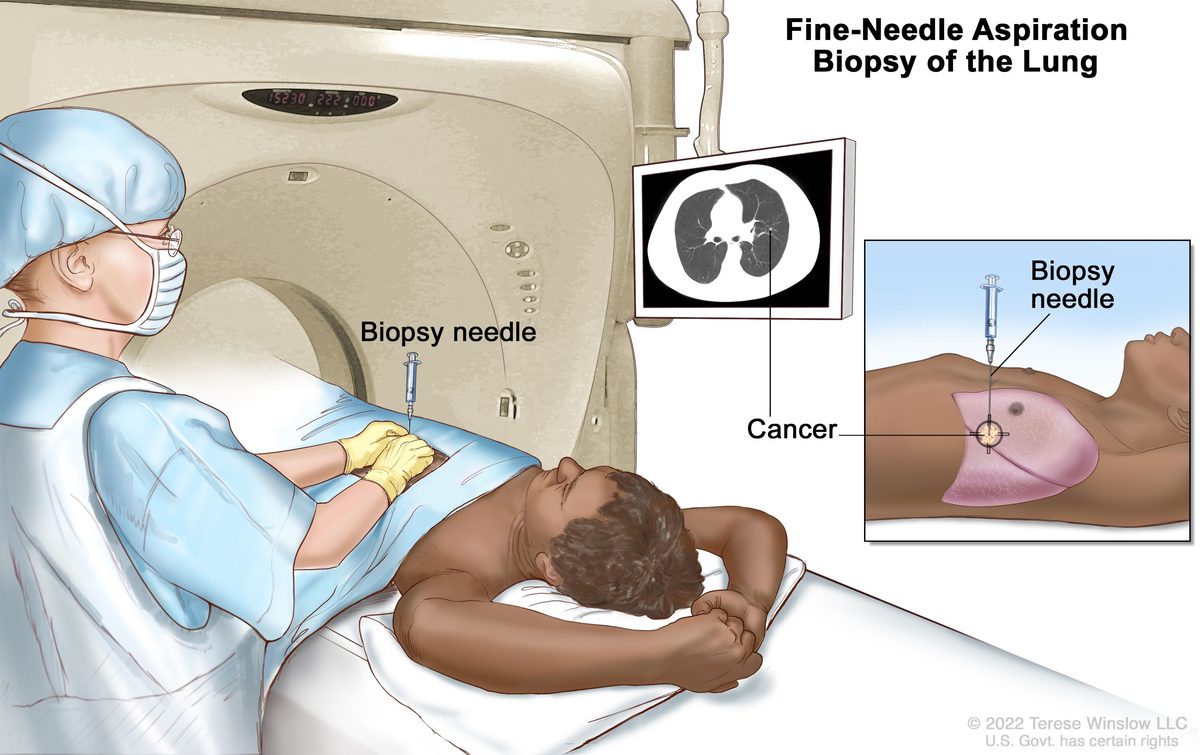
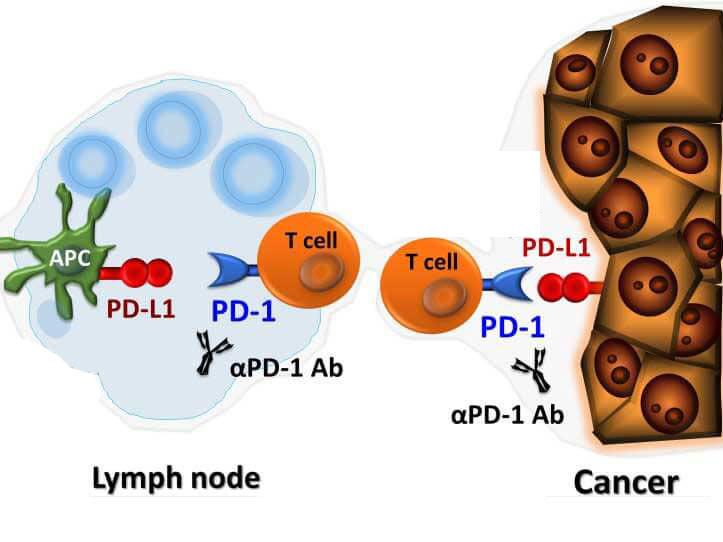
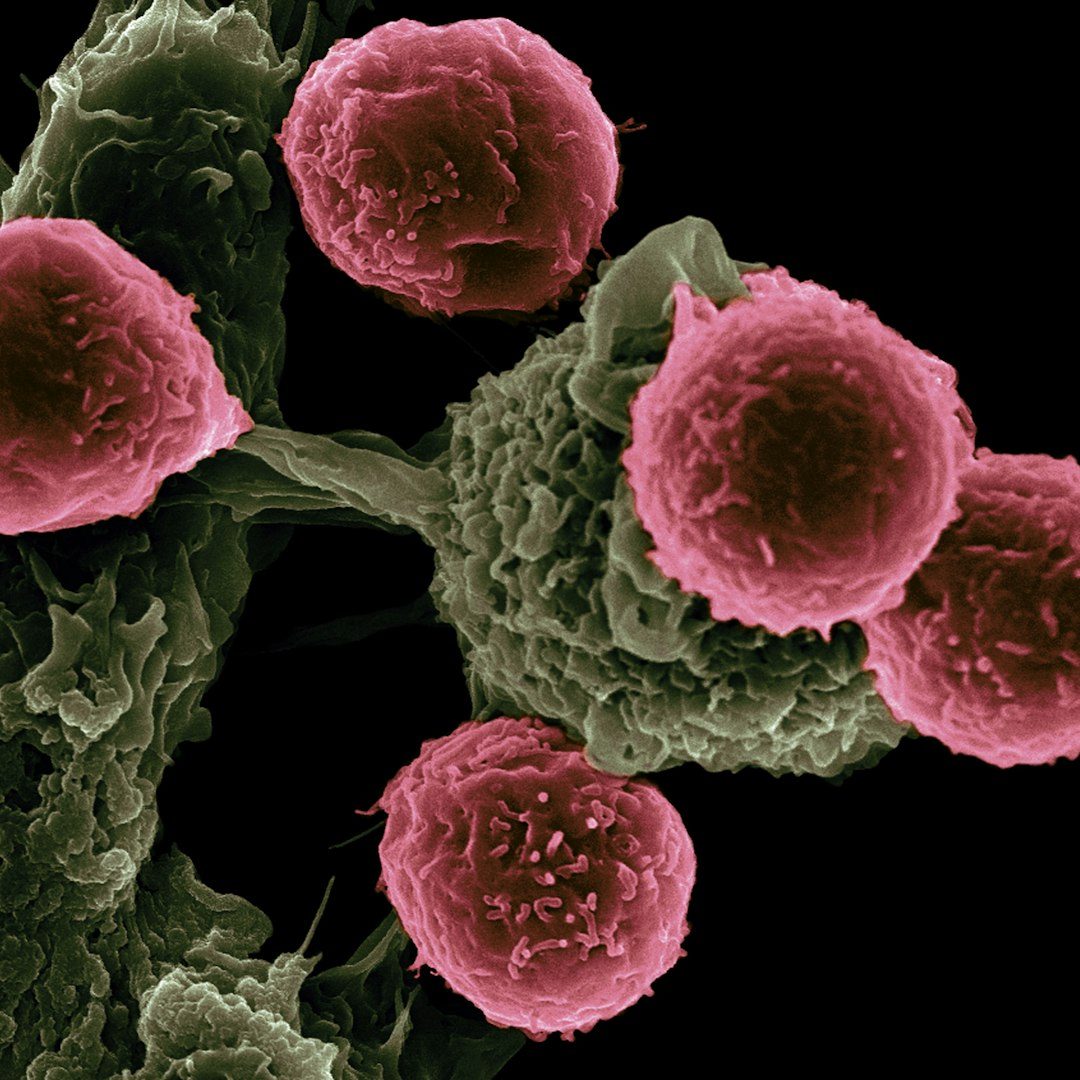
Blinatumomab and the birth of BiTE therapy (2014)
Amgen's blinatumomab (Blincyto) became the first bispecific T-cell engager ever approved, targeting CD19 and CD3 to treat a rare blood cancer called B-cell acute lymphoblastic leukemia. The drug demonstrated that immune cells could be physically redirected to kill cancer cells using an engineered bridging molecule — a concept that had been theorized for decades but never successfully brought to patients.
Blinatumomab provided a new option for patients with a disease that had few effective treatments. It generated $1.6 billion in sales by 2025.
The approval validated the entire BiTE platform and catalyzed a wave of bispecific antibody development across the pharmaceutical industry. It also revealed the platform's key limitation: it initially worked only in blood cancers where immune cells and tumor cells already coexist in the same space.
Tarlatamab's approval represents the BiTE platform's leap from blood cancers into solid tumors — the much larger challenge that blinatumomab's success made possible but could not itself solve.