Cardiac resynchronization therapy transforms heart failure treatment (2001-2005)
2001-2005What Happened
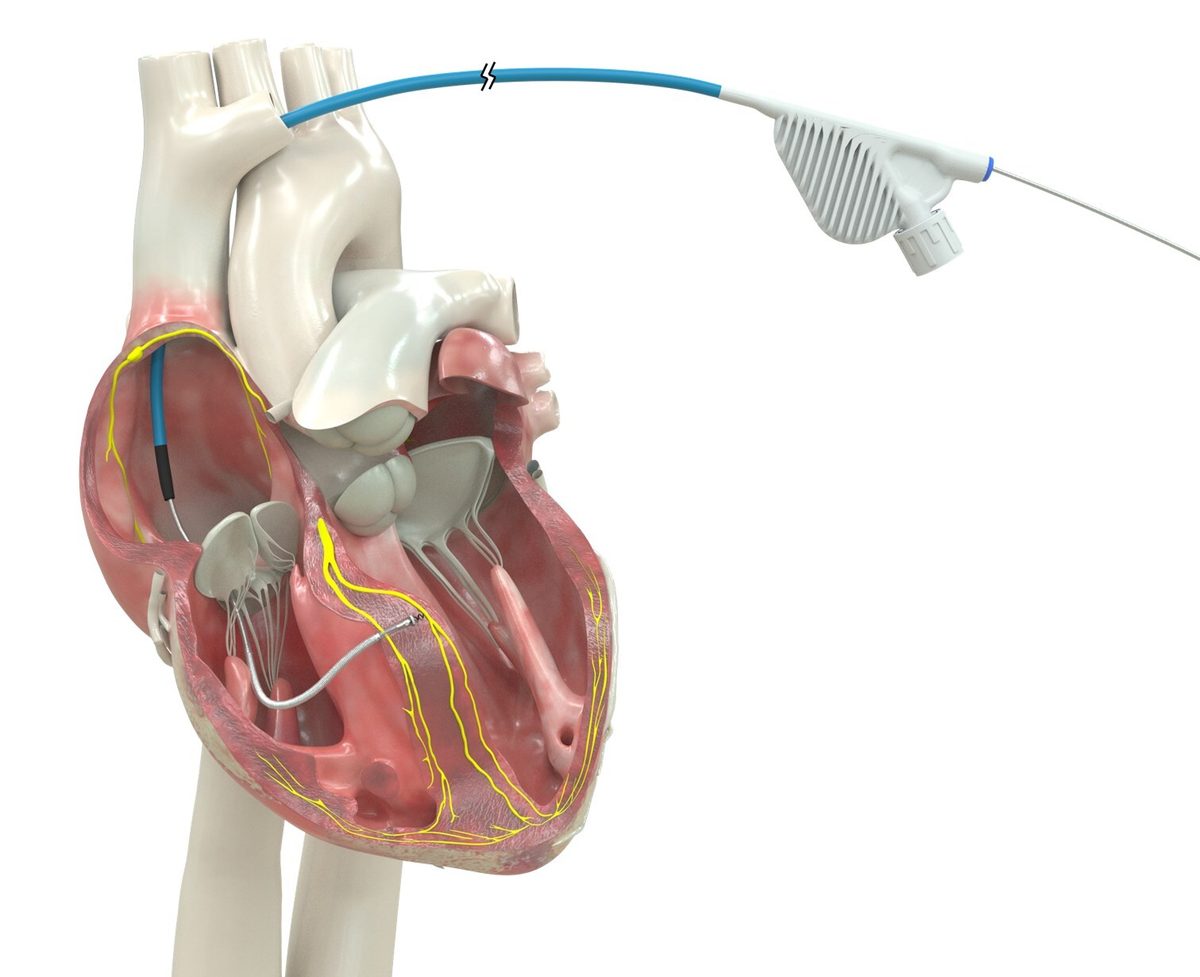
In 2001, the FDA approved the first cardiac resynchronization therapy (CRT) device, which paced both ventricles simultaneously to coordinate their contraction in heart failure patients with electrical conduction delays. The landmark COMPANION trial (2004) and CARE-HF trial (2005) showed CRT reduced death and hospitalization by roughly 36% in appropriate patients, establishing it as standard of care.
Outcome
CRT became the most significant advance in heart failure device therapy, with Medtronic, Boston Scientific, and St. Jude Medical (now Abbott) competing for a market that grew to several billion dollars annually.
CRT proved that how you pace the heart matters as much as whether you pace it — but it still relied on pacing the left ventricle from outside the heart muscle, not through the conduction system. The search for more physiologic approaches continued.
Why It's Relevant Today
The OmniaSecure's conduction system pacing represents the next evolutionary step beyond CRT: instead of pacing both ventricles from outside the muscle, it activates the heart's own wiring. LOT-CRT, also enabled by this approval, combines both approaches.