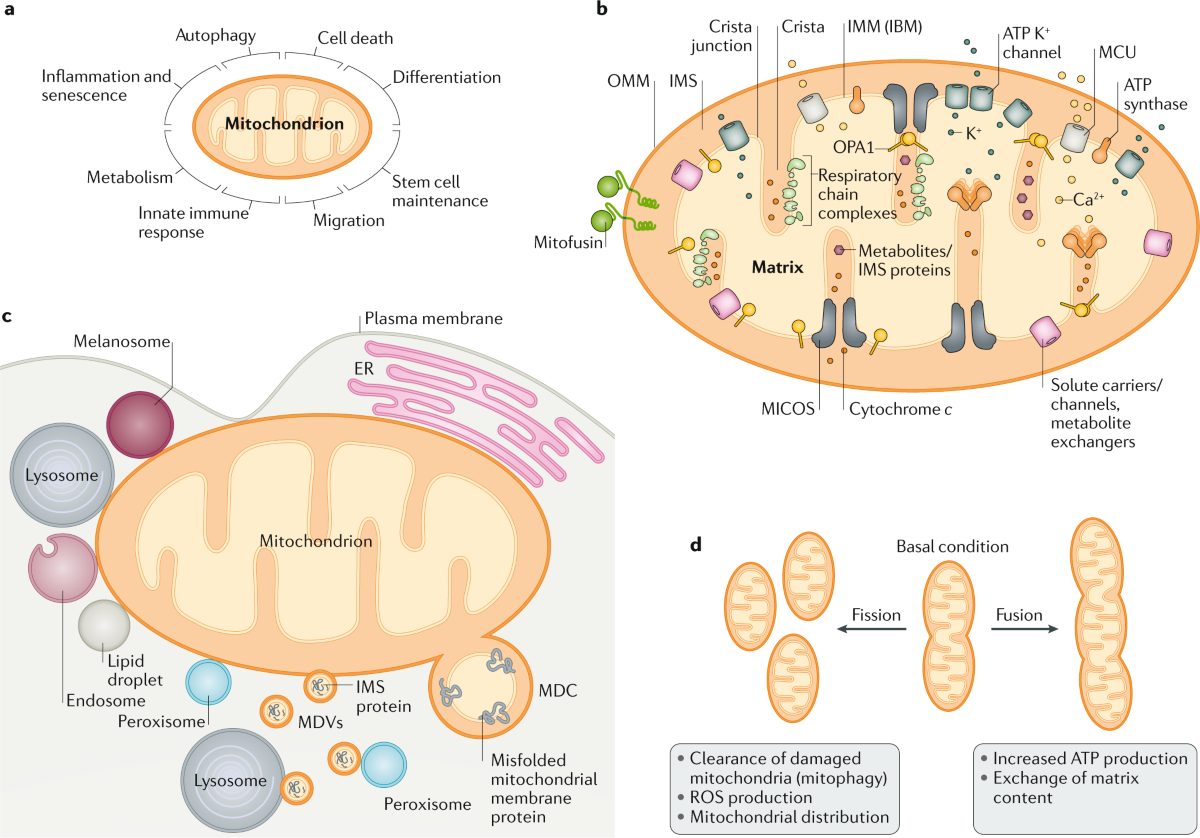
Gene therapy's crash and revival (1990-2023)
1990-2023What Happened
In 1990, four-year-old Ashanthi de Silva became the first gene therapy success, treated for a severe immune deficiency. Nine years later, 18-year-old Jesse Gelsinger died in a University of Pennsylvania gene therapy trial after a massive immune reaction to the viral delivery vehicle — the first publicly identified death in gene therapy. The FDA temporarily halted all U.S. gene therapy trials.
Outcome
Gene therapy research nearly stopped. Public trust collapsed and funding dried up for years.
Researchers spent two decades developing safer delivery vehicles. By 2023, the FDA had approved gene-editing therapies for sickle cell disease, and dozens of gene therapies were in clinical use — but only after the field solved its delivery problem.
Why It's Relevant Today
Mitochondrial transplantation faces a parallel delivery challenge: getting therapeutic cargo into cells safely and efficiently. The capsule technique addresses exactly this bottleneck — much as safer viral vectors eventually unlocked gene therapy's potential.