EU Clinical Trials Directive implementation (2004)
May 2004What Happened
The UK implemented EU Directive 2001/20/EC, applying a single rulebook to all interventional drug trials regardless of risk. Academic and investigator-led research collapsed as costs and bureaucracy multiplied; commercial sponsor activity in the UK fell sharply over the following decade.
Outcome
Trial starts dropped, particularly for non-commercial cancer and paediatric studies. Insurance and sponsorship costs for university-led trials roughly doubled.
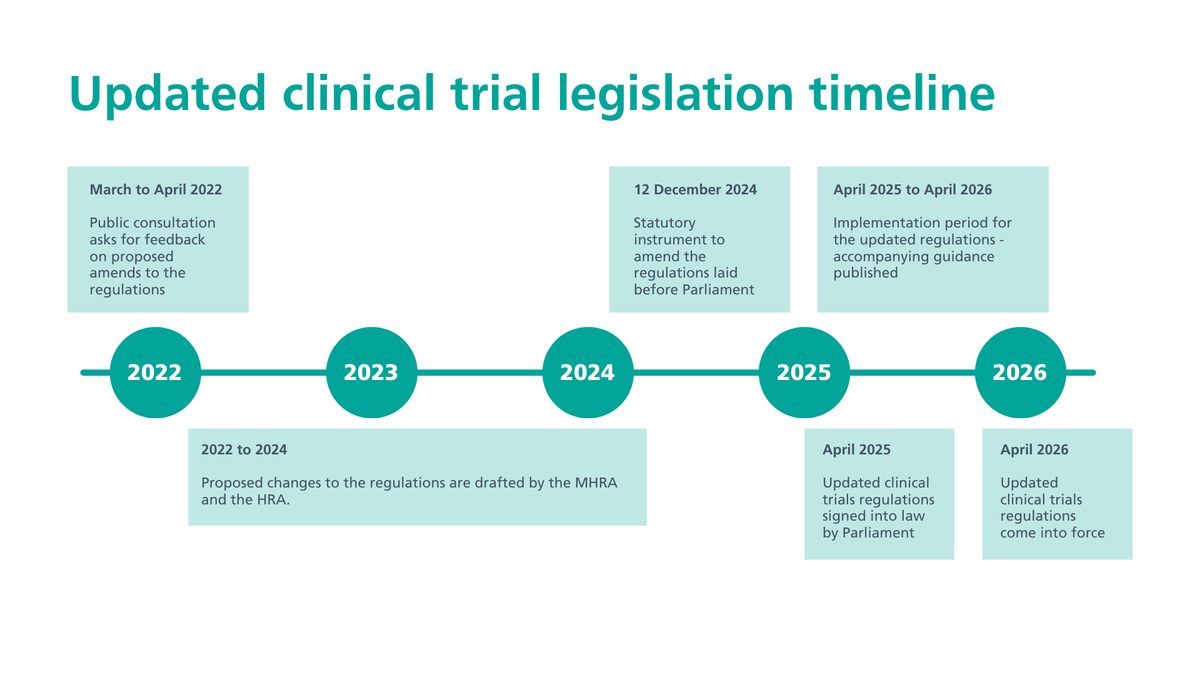
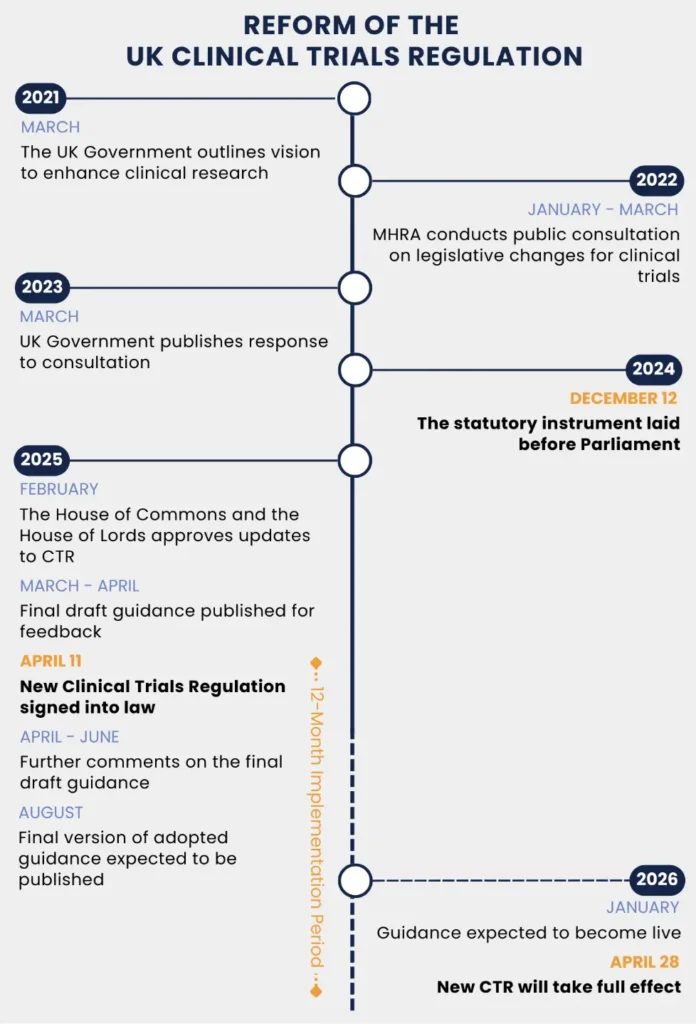
The directive is widely cited as the reason European trial activity migrated to North America and Asia, and it shaped the political case for the post-Brexit reforms that took effect in April 2026.
Why It's Relevant Today
The 2026 reforms are an explicit attempt to fix what the 2004 regime broke—replacing one-size-fits-all review with risk-proportionate routes.