Tofacitinib (Xeljanz) approval for rheumatoid arthritis (2012)
November 2012What Happened
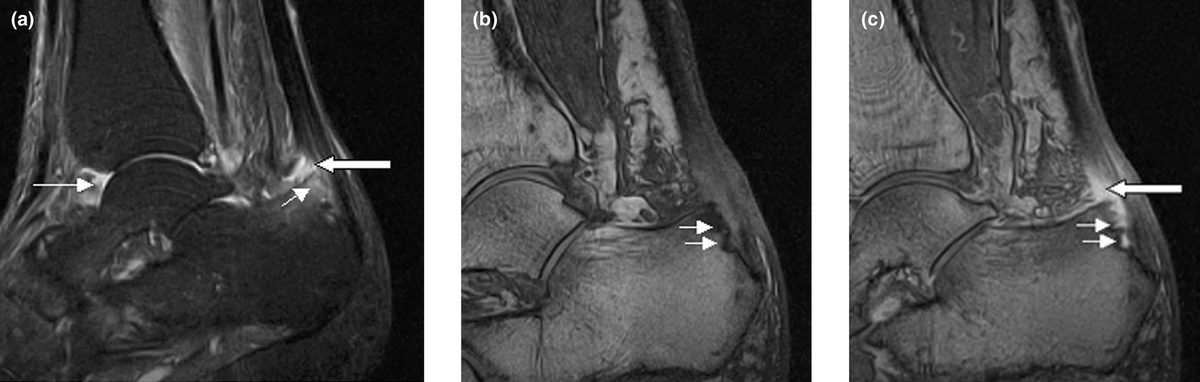
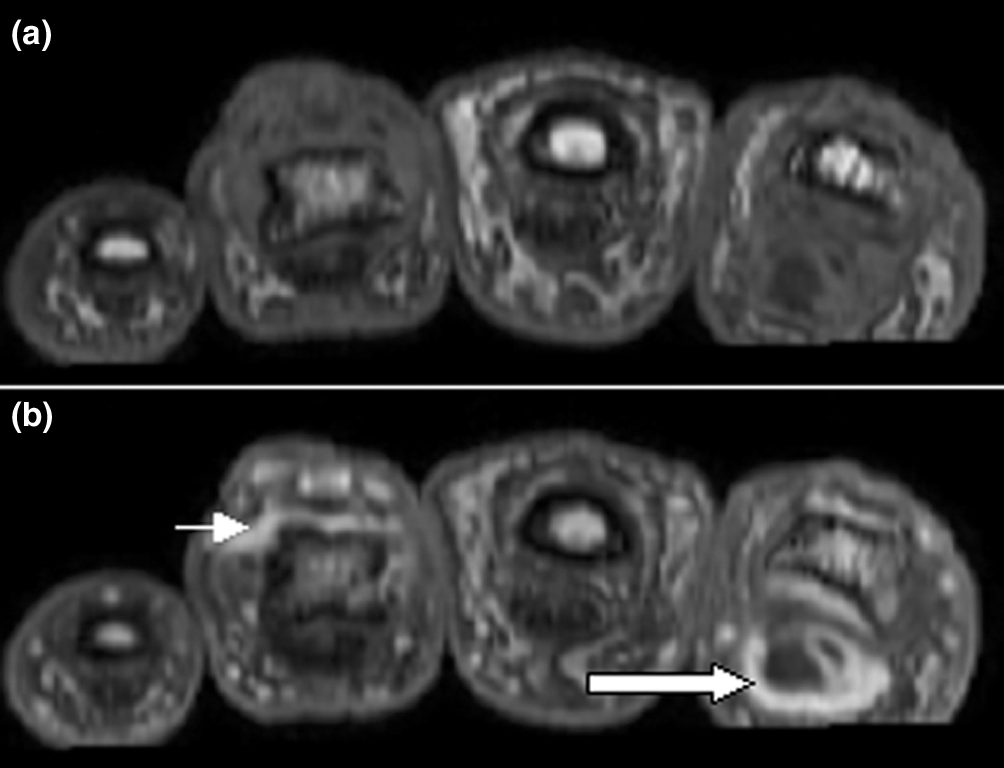
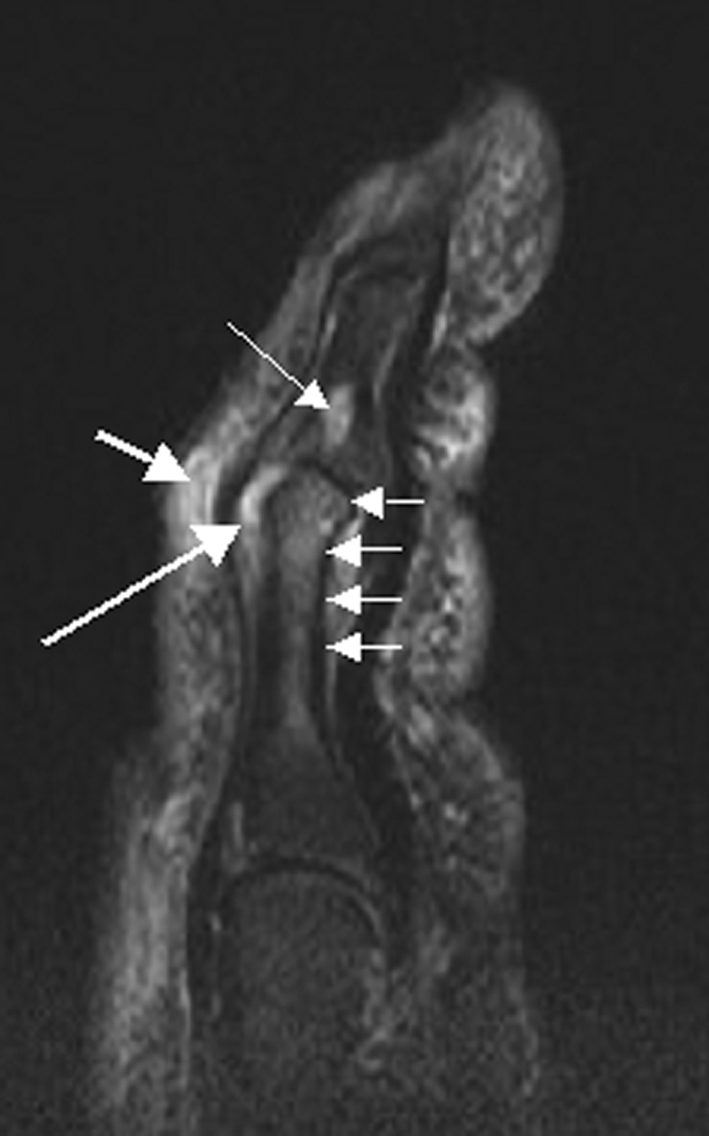
The FDA approved tofacitinib, the first oral Janus kinase inhibitor, for rheumatoid arthritis. Developed by Pfizer, it represented a landmark shift: patients with inflammatory arthritis could take a pill instead of receiving injections of biologic drugs like adalimumab (Humira). Tofacitinib later won approvals for psoriatic arthritis and ulcerative colitis.
Outcome
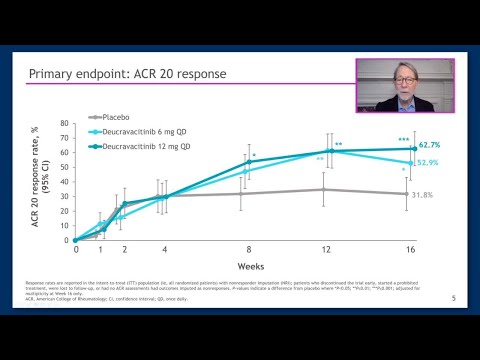
Tofacitinib rapidly gained market share as patients and rheumatologists embraced the convenience of oral dosing. Pfizer projected billions in peak sales.
The 2021 ORAL Surveillance trial revealed increased cardiovascular, cancer, and blood clot risks compared with TNF inhibitors, triggering FDA black box warnings across the entire JAK inhibitor class and dramatically limiting their prescribing. The safety setback created the very market gap that TYK2 inhibitors now aim to fill.
Why It's Relevant Today
Deucravacitinib's path directly echoes tofacitinib's: a first-in-class oral inhibitor expanding from one autoimmune condition into arthritis. But its regulatory story is shaped by the JAK inhibitor safety fallout—TYK2's selective mechanism is specifically designed to avoid the risks that derailed its predecessors.