Rexulti approved as first Alzheimer's agitation treatment (2023)
May 2023What Happened
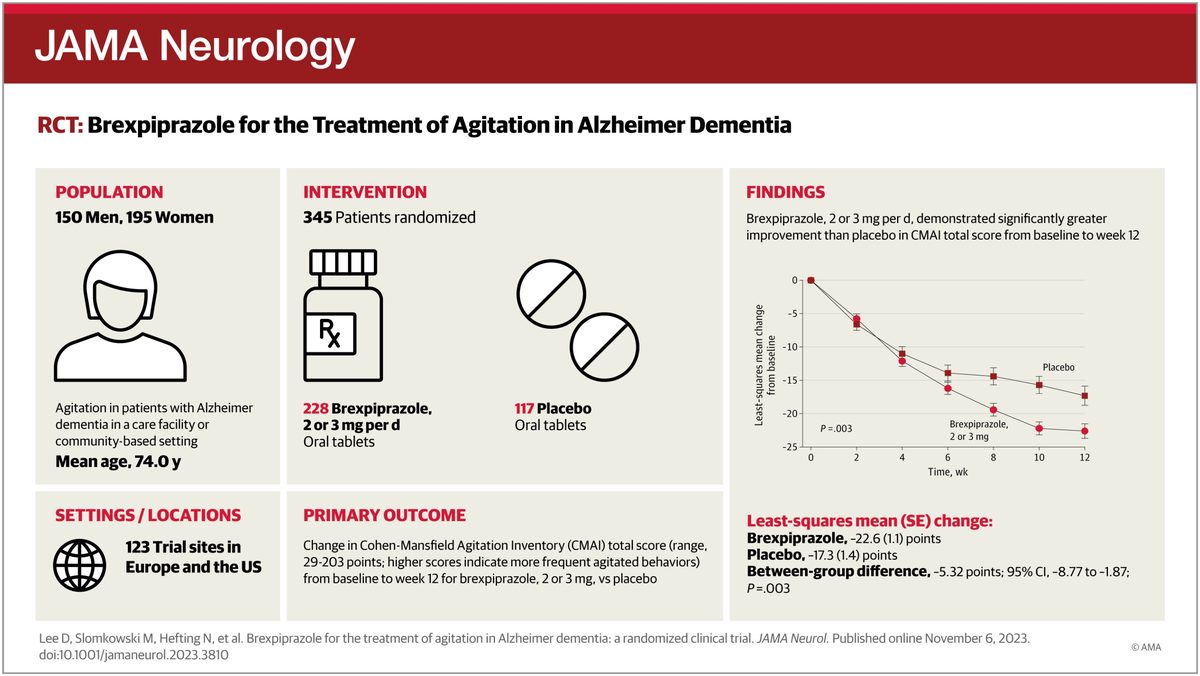
After more than a decade of failed trials by multiple sponsors, the FDA approved Otsuka and Lundbeck's brexpiprazole for agitation in dementia due to Alzheimer's disease. Two pivotal trials showed modest but statistically significant reductions in agitation scores versus placebo.
Outcome
The approval established the first FDA-cleared option in a category where doctors had relied on off-label antipsychotics carrying mortality warnings.
It validated the regulatory pathway for dementia-symptom drugs and incentivized other sponsors—including Axsome—to push their own programs to filing.
Why It's Relevant Today
Rexulti's approval created the category that Auvelity now expands. Without that 2023 precedent, the regulatory and reimbursement frameworks Auvelity steps into would not exist.