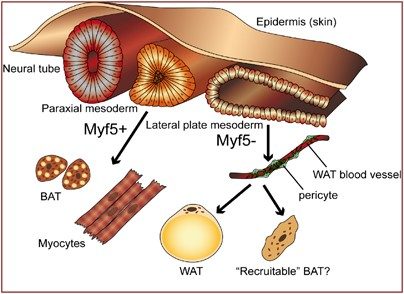
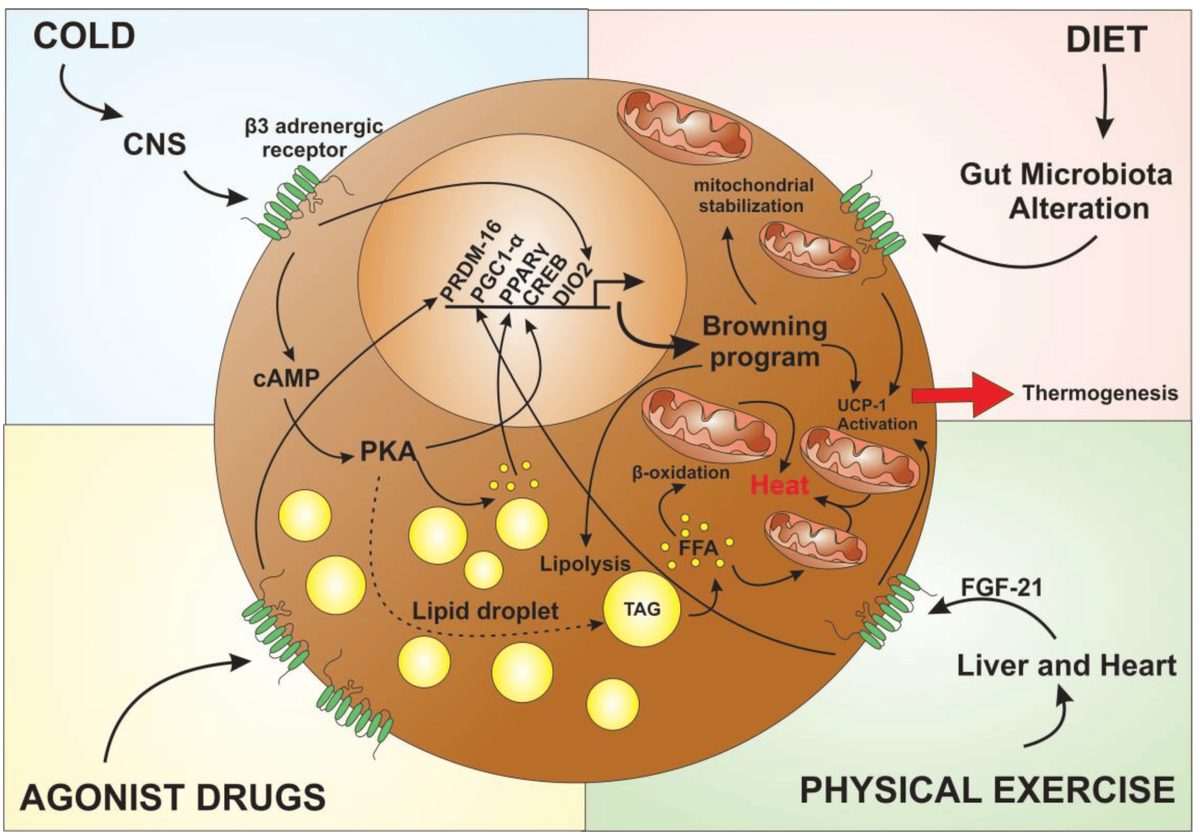
Leptin discovery and the obesity drug disappointment (1994)
December 1994What Happened
Jeffrey Friedman at Rockefeller University cloned the ob gene and its protein leptin, revealing for the first time that fat tissue secretes a hormone regulating body weight. The ob/ob mouse, which lacked leptin, was massively obese; injecting leptin caused dramatic weight loss. Amgen licensed the rights for $20 million and rushed to clinical trials.
Outcome
Leptin administration had minimal effect in most obese humans, who already had high leptin levels and were resistant to it. The drug failed as a general obesity therapy.
Leptin reshaped the entire field's understanding of adipose tissue as an endocrine organ. It eventually found a niche treating rare congenital leptin deficiency. The failure taught researchers that a pathway proven in mice can work entirely differently in obese humans.
Why It's Relevant Today
The SLIT3 discovery faces the same translational question: will a mechanism validated in mice and in human tissue samples hold up when tested as a drug in living obese patients? The 1,500-person tissue analysis is a stronger starting point than leptin had, but the history counsels caution about the gap between biological insight and therapeutic impact.