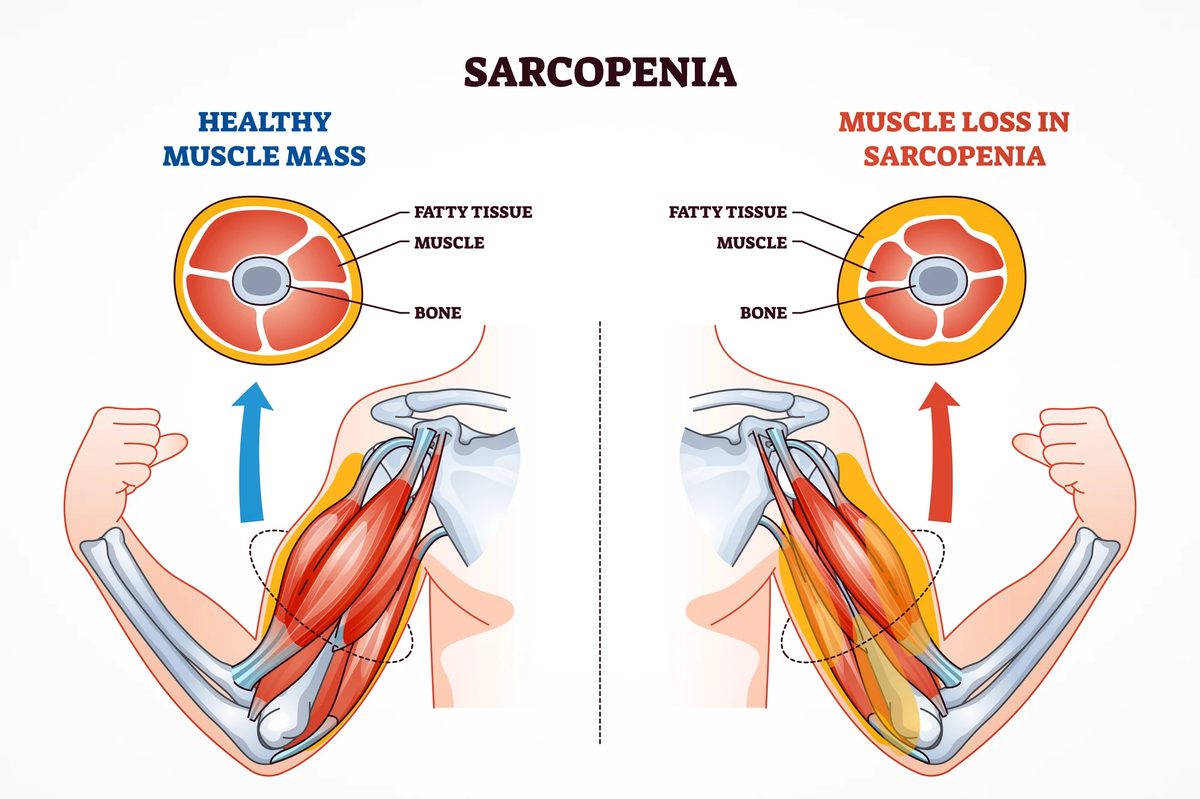
The biggest knock against today's blockbuster weight-loss drugs is that they burn muscle along with fat — up to 40% of the weight a patient loses on semaglutide can be lean tissue like muscle and bone. A new class of therapies now entering clinical trials attacks obesity through entirely different biology, targeting genes and metabolic pathways that strip fat while leaving muscle intact. On March 26, 2026, Wave Life Sciences reported that a single injection of its RNA-silencing drug WVE-007 cut visceral fat by 14.3% over six months while patients actually gained 2.4% lean mass — a profile no existing obesity drug can match.
The biggest knock against today's blockbuster weight-loss drugs is that they burn muscle along with fat — up to 40% of the weight a patient loses on semaglutide can be lean tissue like muscle and bone. A new class of therapies now entering clinical trials attacks obesity through entirely different biology, targeting genes and metabolic pathways that strip fat while leaving muscle intact. On March 26, 2026, Wave Life Sciences reported that a single injection of its RNA-silencing drug WVE-007 cut visceral fat by 14.3% over six months while patients actually gained 2.4% lean mass — a profile no existing obesity drug can match.
The result marks one of several emerging approaches that could reshape a market Goldman Sachs projects will reach $95–130 billion by 2030. Arrowhead Pharmaceuticals is targeting the same liver gene with its own RNA therapy and has shown that combining it with tirzepatide doubles weight loss. Rivus Pharmaceuticals is testing an oral pill that accelerates metabolism in fat cells specifically. If any of these mechanisms prove safe and effective at scale, they could either complement GLP-1 drugs or compete with them directly — redefining what 'weight loss' means in clinical practice from simple pounds lost to the quality of tissue that remains.