Keytruda, the world's best-selling drug, generated $31.7 billion for Merck in 2025 — roughly 40% of the company's total revenue. Its key U.S. patents expire in 2028, and when they do, cheaper copies will enter the market and that revenue will begin to evaporate. Merck has now spent over $25 billion on three acquisitions in nine months to build the portfolio that will need to replace it.
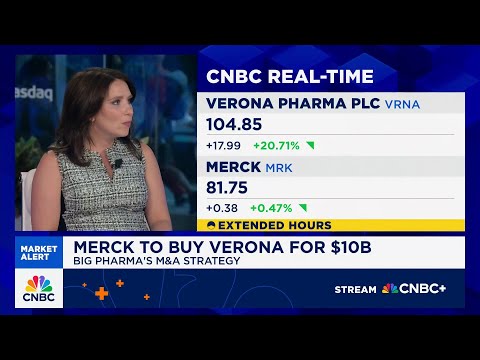
The latest move, a $6.7 billion deal for Terns Pharmaceuticals announced March 25, gives Merck an experimental leukemia pill that analysts believe could eventually rival Novartis's Scemblix, which is on track to become a multibillion-dollar franchise. Combined with a $10 billion buy for respiratory drugmaker Verona Pharma and a $9.2 billion deal for flu-drug developer Cidara Therapeutics, Merck is executing one of the most aggressive pipeline-rebuilding campaigns in pharmaceutical history — a direct response to what may be the largest single-drug patent cliff the industry has ever faced.