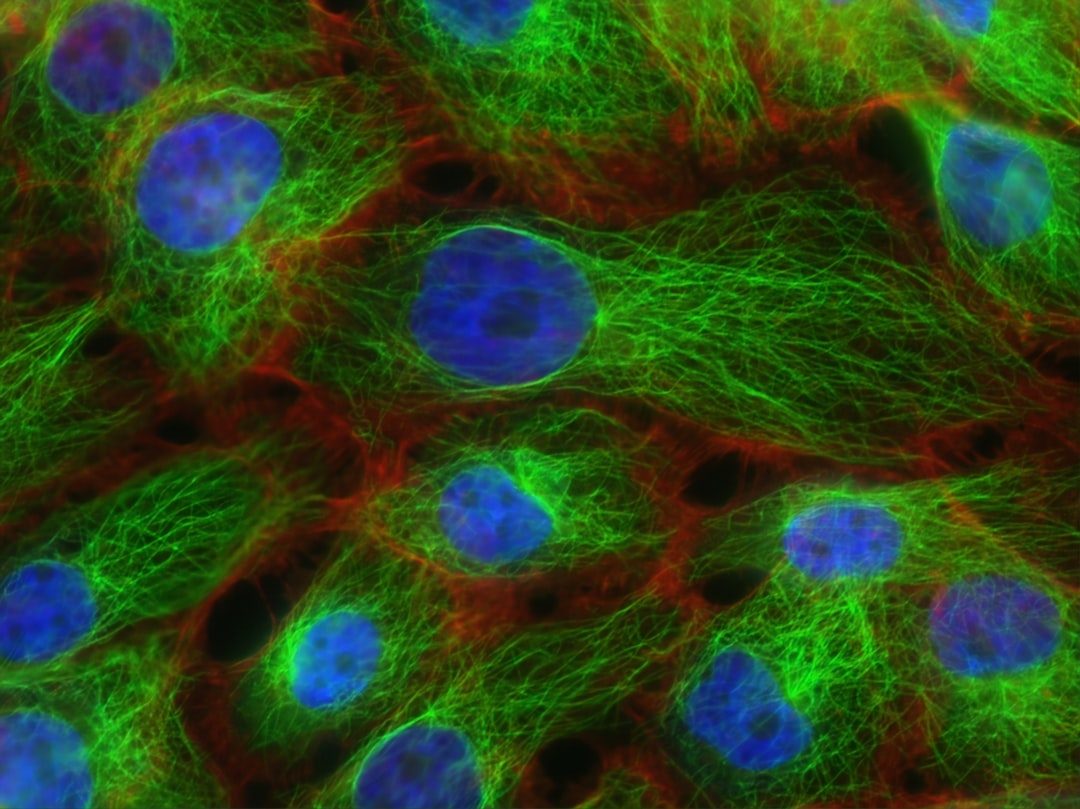
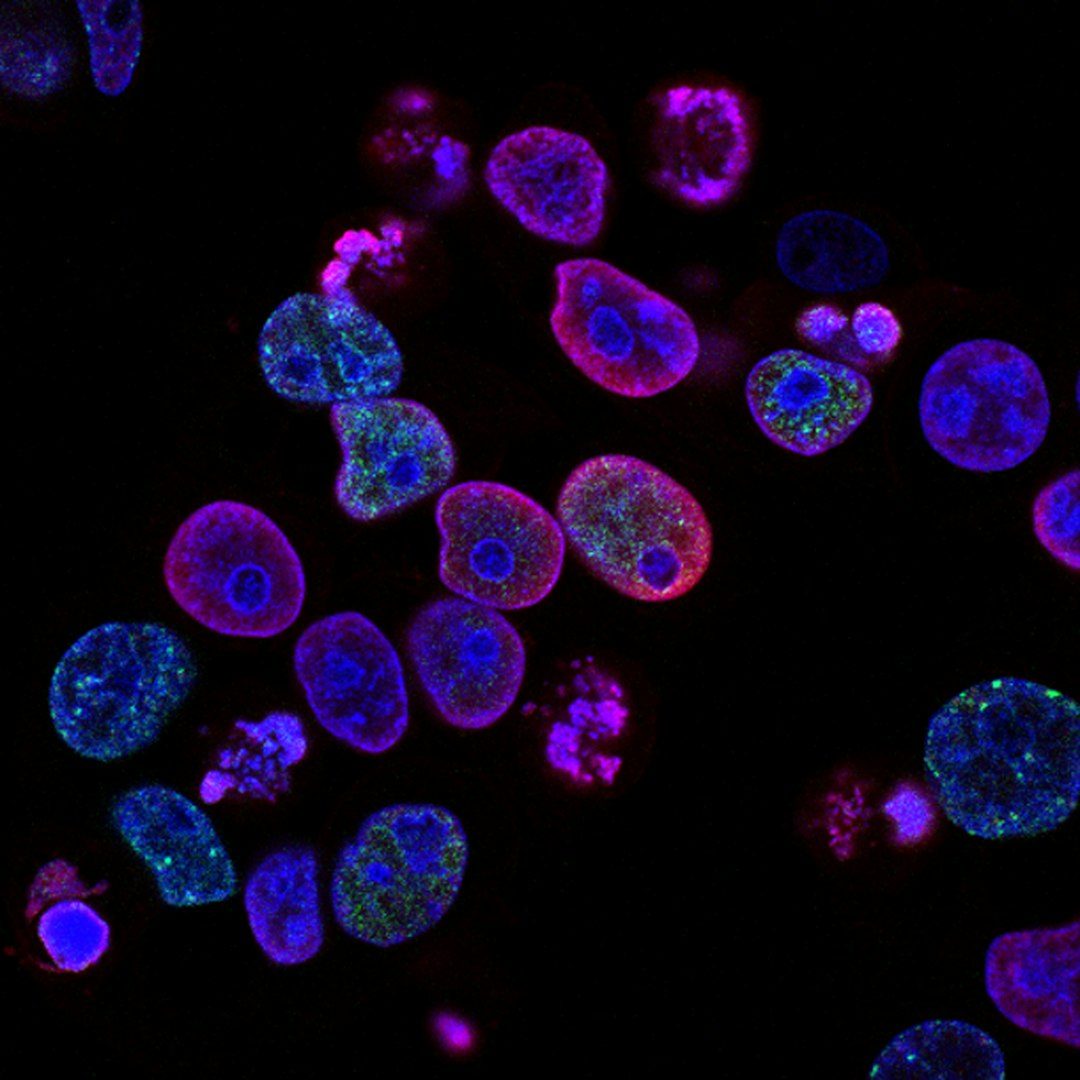
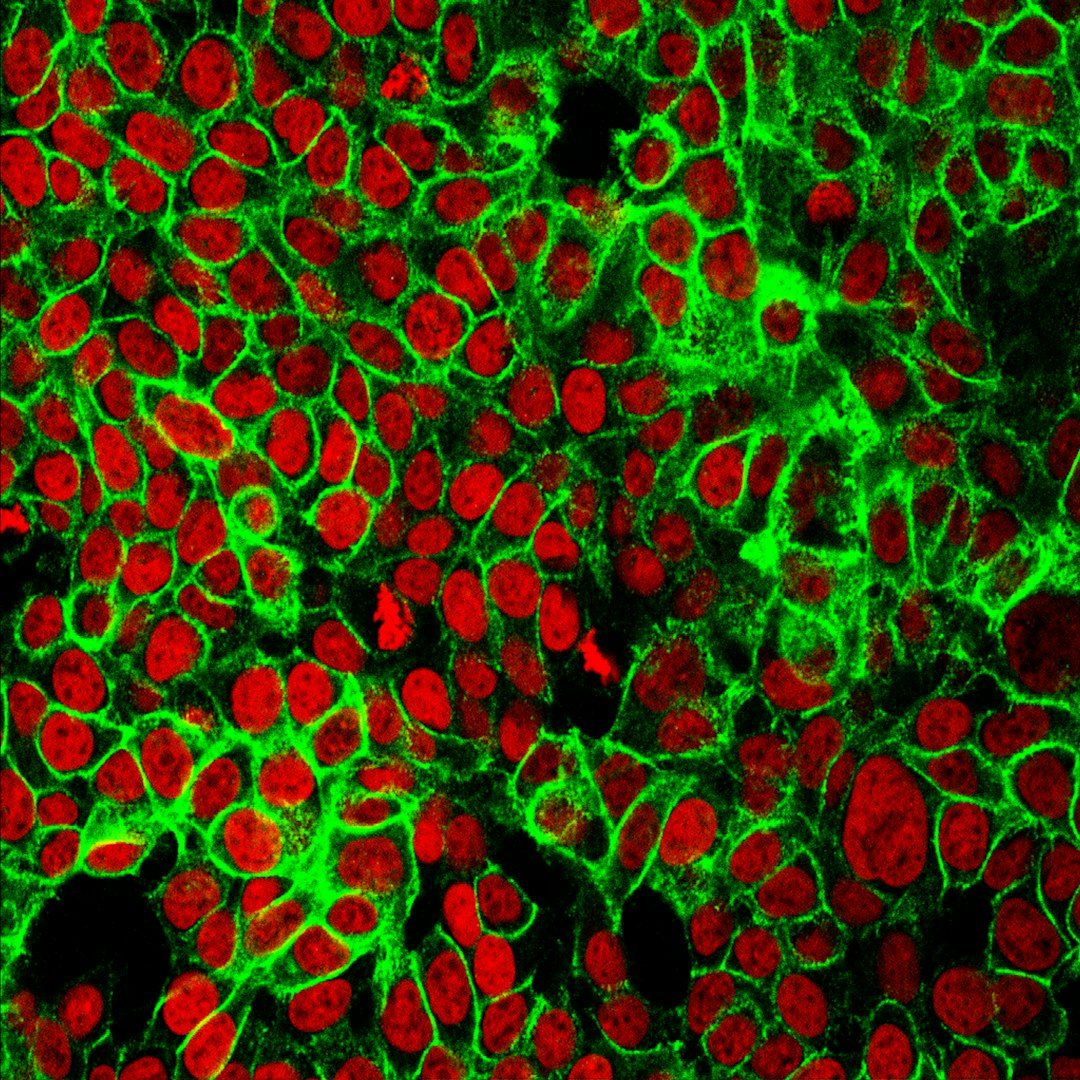
For decades, biologists treated the cell's energy-producing machinery and its DNA-reading machinery as separate systems operating in separate compartments. A study published March 6 in Nature Communications upends that assumption: more than 200 metabolic enzymes, many of them normally associated with energy production in mitochondria, are physically attached to human DNA inside the nucleus. About 7% of all proteins bound to chromatin turn out to be metabolic enzymes, forming what the researchers describe as a 'mini metabolism' within the nucleus itself.
For decades, biologists treated the cell's energy-producing machinery and its DNA-reading machinery as separate systems operating in separate compartments. A study published March 6 in Nature Communications upends that assumption: more than 200 metabolic enzymes, many of them normally associated with energy production in mitochondria, are physically attached to human DNA inside the nucleus. About 7% of all proteins bound to chromatin turn out to be metabolic enzymes, forming what the researchers describe as a 'mini metabolism' within the nucleus itself.
The finding matters because each tissue and cancer type displays its own distinct pattern of nuclear enzymes, a signature the team calls a 'nuclear metabolic fingerprint.' Oxidative phosphorylation enzymes, for instance, are abundant on the chromatin of breast cancer cells but largely absent from lung cancer cells. That tissue-specific variation opens a new front for cancer diagnostics and targeted therapies, and raises a fundamental question: how do enzymes far too large for the nucleus's usual import channels get inside?