Trastuzumab and the birth of HER2-targeted therapy (1998)
September 1998What Happened
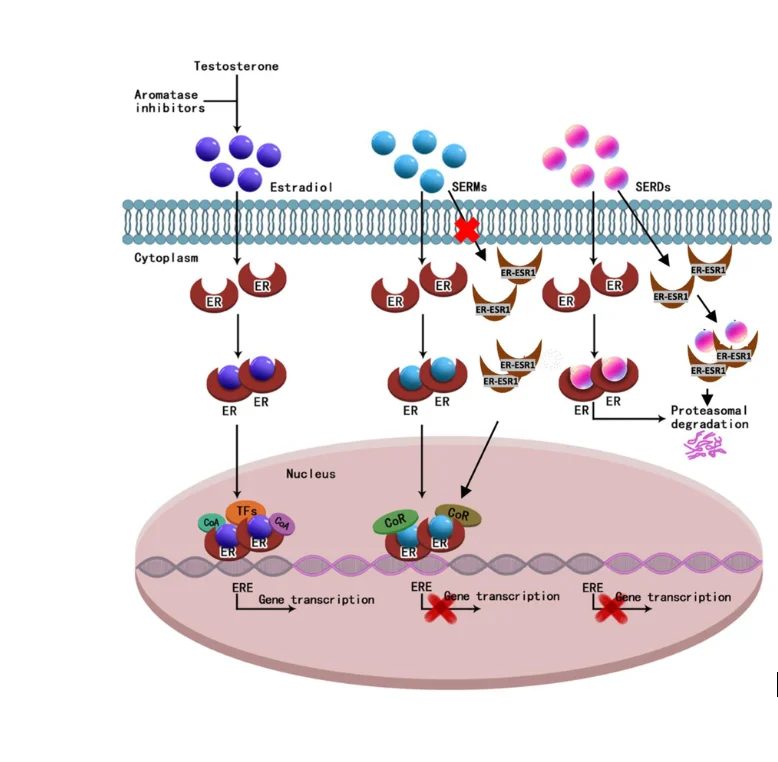
FDA approved Genentech's Herceptin for the 25-30% of breast cancer patients whose tumors overexpress the HER2 protein, paired with a companion diagnostic test. It was the first time a cancer drug was approved alongside a biomarker test that selected which patients should get it.
Outcome
Established HER2 status as a routine pathology test and gave HER2-positive patients — previously the worst-prognosis group — the best long-term outcomes in breast cancer.
Became the template for every targeted-therapy-plus-companion-diagnostic approval that followed: EGFR in lung cancer, BRAF in melanoma, BRCA in ovarian cancer.
Why It's Relevant Today
Camizestrant could extend that template by adding a temporal dimension — not just selecting patients at diagnosis based on tumor tissue, but switching therapy mid-course based on what blood reveals about evolving resistance.