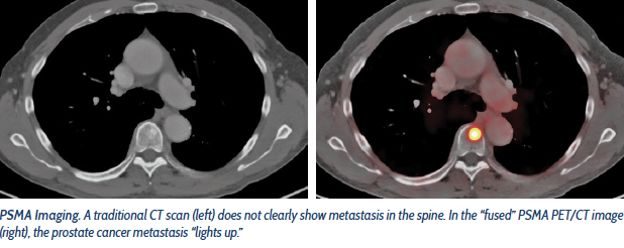
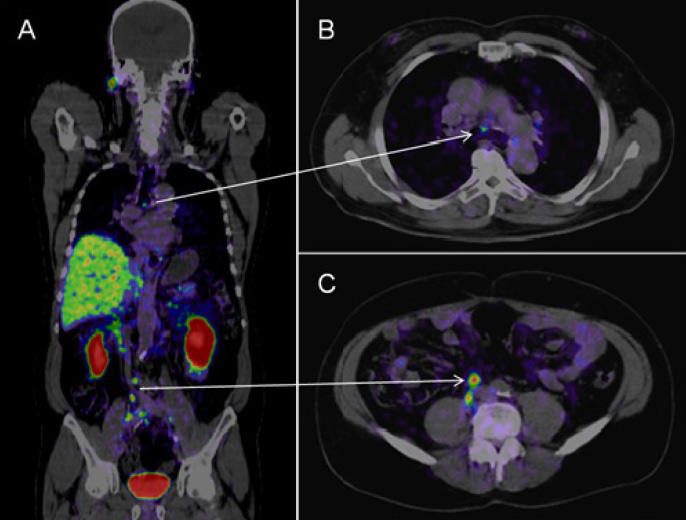
MRI replaces blind biopsies for prostate cancer (2017-2021)
2017-2021What Happened
The PRECISION trial in 2018 and the STHLM3-MRI trial in 2021 demonstrated that magnetic resonance imaging could be used to target prostate biopsies rather than sampling the gland blindly. The STHLM3 trial, enrolling over 12,000 men in Stockholm, showed that combining a risk-prediction blood test with MRI reduced overdiagnosis while maintaining detection of dangerous cancers.
Outcome
MRI-targeted biopsy became the standard of care in many countries, reducing the number of men biopsied by roughly 48% and cutting detection of harmless cancers by 62%.
MRI set the precedent that imaging could safely replace some biopsies, but it still left a large group of men with ambiguous results who were sent for biopsy anyway. PRIMARY2 addresses exactly this gap.
Why It's Relevant Today
PRIMARY2 picks up where MRI-targeted biopsy left off. The trial specifically enrolled men whose MRI results were inconclusive, showing that adding PSMA PET to the diagnostic pathway can resolve ambiguity that MRI alone cannot.