Enhertu redefines HER2-positive cancer (2019-2022)
December 2019 to August 2022What Happened
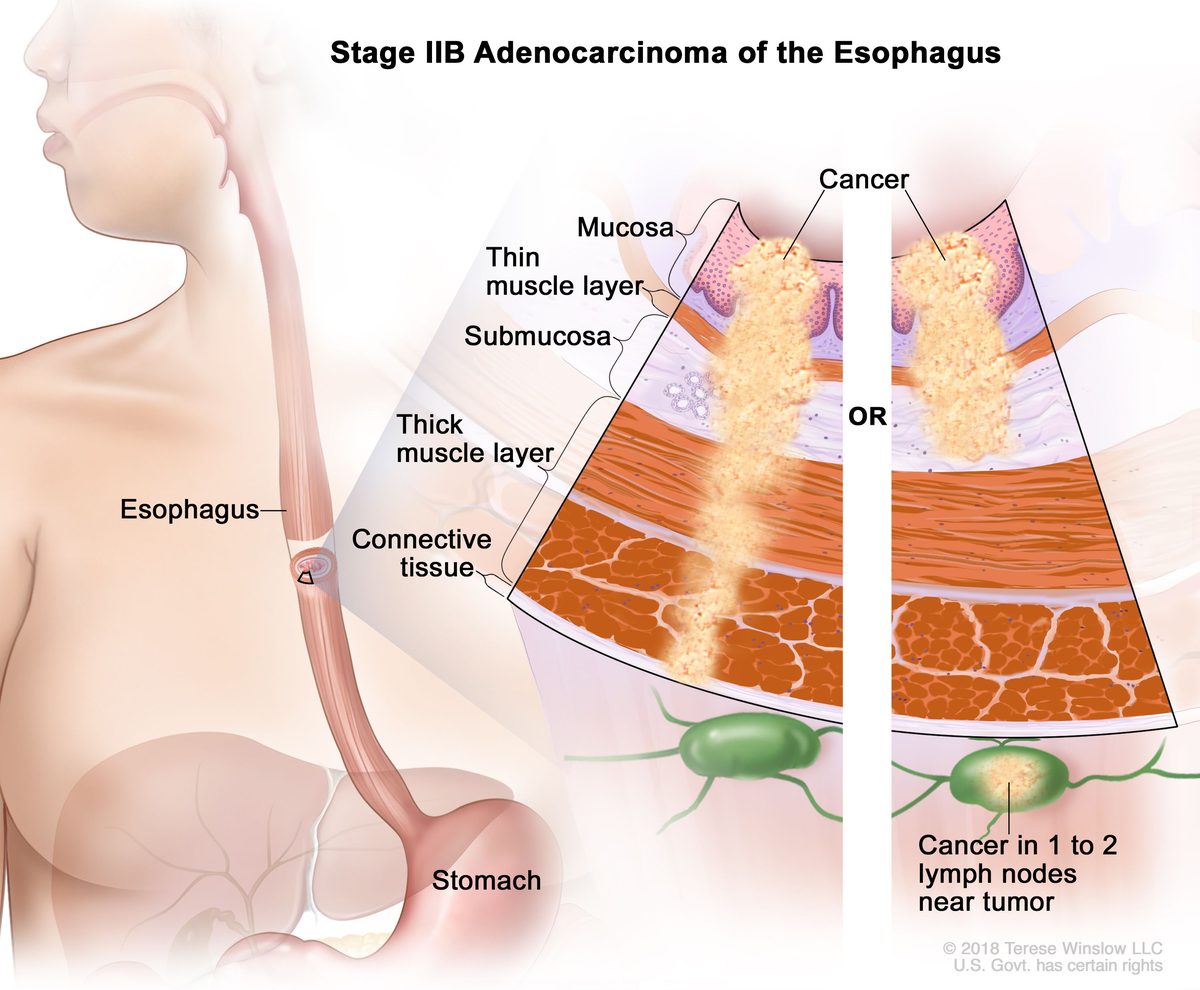
Daiichi Sankyo and AstraZeneca won FDA approval for trastuzumab deruxtecan, branded Enhertu, in HER2-positive breast cancer. Follow-up trials showed it also worked in HER2-low breast cancer and HER2-positive gastric and lung cancers, expanding the addressable population sharply.
Outcome
Enhertu sales passed $1 billion in 2022 and the drug became standard of care across multiple HER2 settings.
The success validated DXd-payload antibody-drug conjugates as a platform and pulled massive capital into the ADC space, including the Chinese programs now reaching late-stage trials.
Why It's Relevant Today
HS-20093 uses the same class of payload chemistry against a different tumor antigen. If B7-H3 expression patterns play out like HER2 did, the drug could move from late-line salvage into earlier treatment settings.