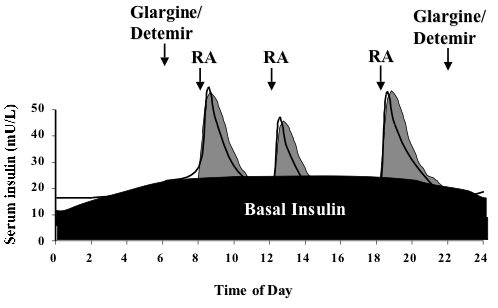
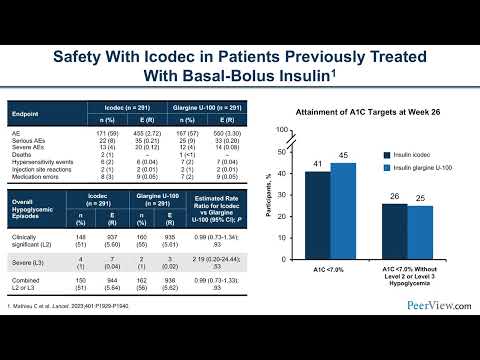
Weekly insulin becomes standard of care for new type 2 diabetes patients starting basal insulin
If Awiqli's pricing is competitive with existing long-acting daily insulins and insurance formularies grant broad coverage, the convenience advantage could make weekly insulin the default starting point for the millions of type 2 diabetes patients who initiate basal insulin each year. The 93.7% patient preference rate in clinical trials suggests rapid adoption once access barriers are resolved. Eli Lilly's efsitora alfa entering the market would accelerate this shift by creating price competition.